Cagrilintide + Semaglutide Stack: How Wholesale Buyers Plan Dual-Peptide Portfolios
Cagrilintide + Semaglutide: Planning a Dual-Peptide GLP-1 Stack
Many wholesale buyers who start with single-molecule GLP-1 projects eventually ask the same question: what happens if we combine appetite-support from two different pathways? That is where a fixed dual-peptide kit such as Cagrilintide 5 mg + Semaglutide 5 mg becomes interesting for portfolio planning.
Different Roles in the Same Stack
In most project designs, Semaglutide is treated as the GLP-1 backbone, while Cagrilintide is used to tighten satiety and caloric-intake control:
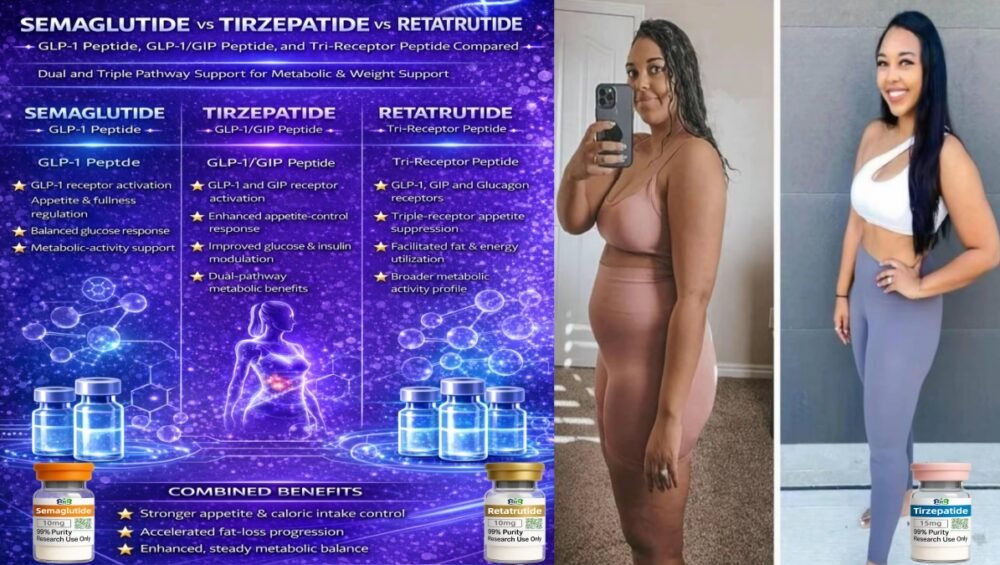
- Semaglutide: GLP-1 receptor activation, appetite signalling, balanced glucose response, and overall metabolic-activity support.
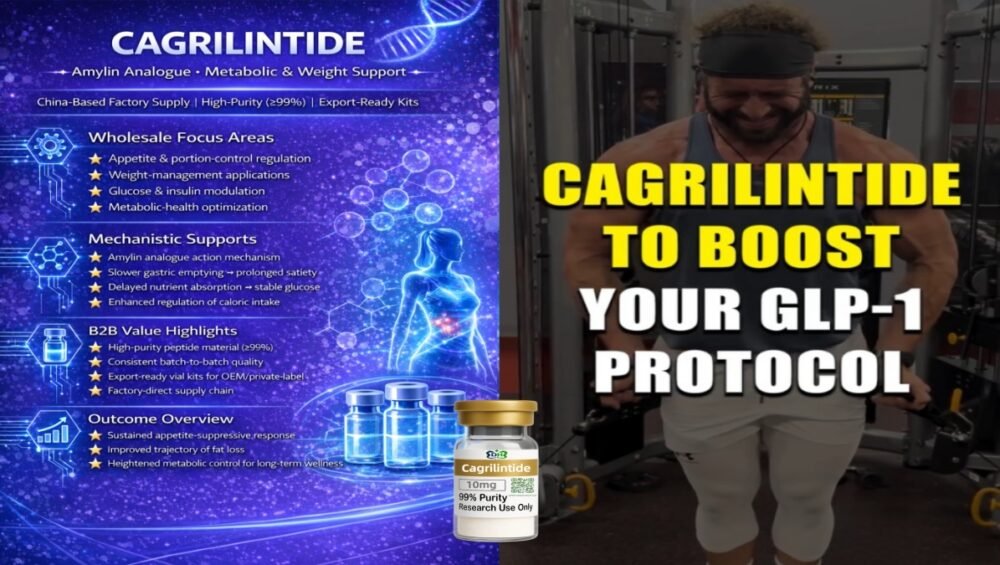
- Cagrilintide: slower gastric emptying, prolonged fullness and smoother caloric-intake patterns.
By sourcing both from the same China-based factory – for example Semaglutide peptide kits plus dedicated Cagrilintide kits – buyers can explore each molecule separately before locking in a fixed dual-peptide protocol.
Why Some Buyers Move Toward a Dual-Peptide Approach
There are a few recurring reasons why B2B customers add a Cagrilintide + Semaglutide stack to their GLP-1 portfolio:
- Existing GLP-1 projects show good response, but teams want stronger appetite-control signals and better day-to-day satiety stability.
- OEM and private-label brands are looking for a premium “enhanced satiety” or “next-step” program above standard GLP-1 options.
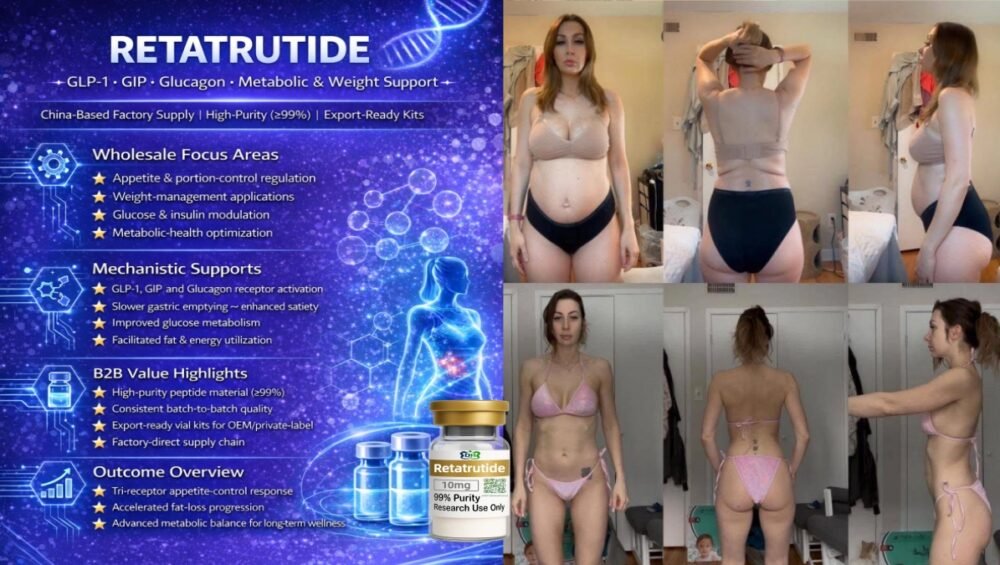
- Internal R&D groups want to understand how amylin-pathway support changes long-term weight-management trajectories.
Instead of running two fully separate supply chains, a single factory-made kit with pre-defined strengths is often easier to standardise and document.
Typical Study and Portfolio Scenarios
In practice, buyers rarely jump straight into dual-peptide work without preparation. A common roadmap looks like this:
- Start with Semaglutide-only projects to understand GLP-1 behaviour and documentation requirements.
- Add single-molecule Cagrilintide kits into pilot models that focus on satiety and caloric-intake tracking.
- Once internal data is consistent, migrate to a fixed dual-peptide format such as Cagrilintide 5 mg + Semaglutide 5 mg vials for larger cohorts or OEM roll-outs.
This step-by-step approach makes it easier to separate the contribution of each peptide, while still ending with a simple kit format for export and routine re-ordering.
Operational Considerations for Dual-Peptide Kits
From a logistics point of view, dual-peptide projects benefit from the same discipline as single-molecule work:
- Use clear batch coding and keep GLP-1 and amylin components traceable in your records.
- Align storage conditions (2–8 °C, dry and dark) for all related vials and reference standards.
- Keep COA, HPLC data and other quality documentation accessible for both Cagrilintide and Semaglutide components, especially for OEM and regulatory reviews.
Because Cagrilintide + Semaglutide kits bundle both peptides into a single export-ready format, many teams find day-to-day handling easier than working with separate suppliers.
How to Position the Stack Inside a Wider Weight-Loss Catalog
From a commercial planning angle, dual-peptide kits are usually positioned as an advanced option next to classical GLP-1 products. A typical catalog structure looks like:
- Core GLP-1 range – e.g. standard Semaglutide peptide kits.
- Optimisation tools – stand-alone Cagrilintide kits for satiety-focused research.
- Premium stacks – fixed dual-peptide formats such as Cagrilintide + Semaglutide vials for projects that already understand both molecules.
This layered approach keeps entry-level projects simple, while still giving more advanced teams a clear upgrade path inside the same China-based factory ecosystem.
Related Products
- Cagrilintide 5 mg + Semaglutide 5 mg – Dual Peptide GLP-1 Stack
- Cagrilintide Peptide Kits – Amylin Analogue for Satiety-Focused Projects
- Semaglutide Peptide Kits – GLP-1 Backbone for Weight-Loss Portfolios
Compliance Notice
THIS CONTENT IS PROVIDED FOR LABORATORY AND R&D DISCUSSION ONLY.
ALL PRODUCTS ARE INTENDED AS RESEARCH CHEMICALS AND ARE NOT LABELED FOR HUMAN USE.